With each new generation of electronics, components and systems get smaller and more powerful. Many of the advantages realized with 01005 components come down to size—signals travel shorter distances within devices leading to faster performance speeds. The aerospace industry was one of the first to demand miniaturization as lighter weight, smaller electrical components propelled aircraft efficiency and reduced material costs. Across all sectors, cost savings are being derived from the lighter weight and smaller sizing of devices engineered with 01005 packages.
Increasingly, original equipment manufacturers (OEMs) are utilizing miniaturized components to improve the competitiveness of their already existing product offerings and deliver entirely new, often disruptive, products. It is predicted that the medical devices sector will grow 38% from 2015 to 2022, primarily due to increased integration between smartphones, wearables and connected health solutions. Let’s examine more closely the advantages OEMs in the medical device sector are realizing due to component miniaturization.
Biosensing Medical Devices & Miniaturized Components
Biosensor and medical device designers are looking to push the limits of electronics manufacturing. Implantable biosensing devices and diagnostic products require circuits that must bend in new ways and printed circuit boards (PCBs) that must deliver added functionality in smaller, tighter spaces. New approaches, materials, pieces of equipment and inventive thinking must occur for medical device manufacturers to realize the advantages of miniaturized components. The biggest challenge OEMs developing biosensing and smart medical devices face is reducing the need for surgical device replacements and increasing health complications.
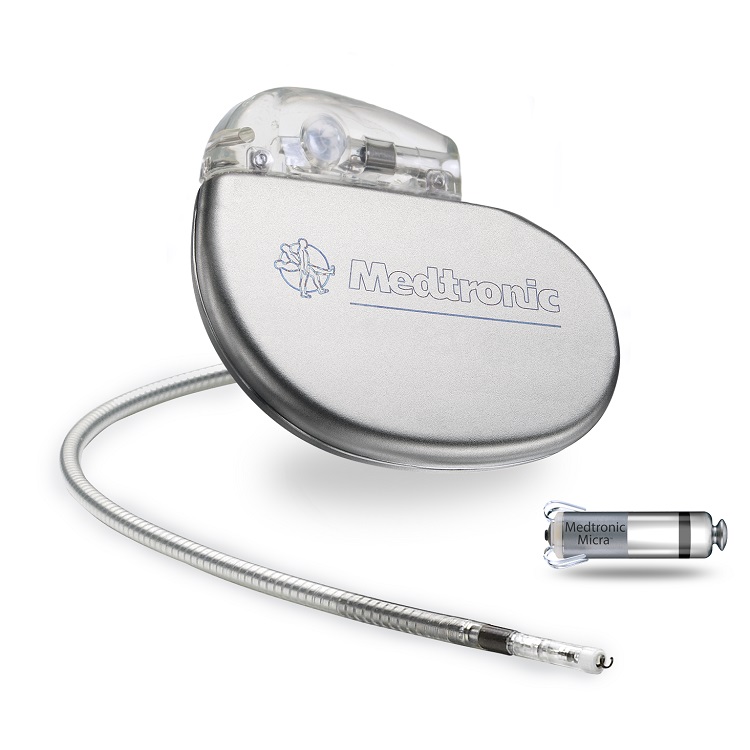 Standout Innovation | Medtronic Micra: The world’s smallest pacemaker, released this past spring, is the result of a “deep miniaturization” manufacturing initiative lasting ten years. Unlike other pacemakers, this revolutionary piece of implantable medical equipment is implanted inside of the heart. Researchers have found that this exceptionally small product is capable of industry-leading performance. When compared to traditional pacing systems, Medtronic’s miniaturized device caused significantly fewer major health complications.
Standout Innovation | Medtronic Micra: The world’s smallest pacemaker, released this past spring, is the result of a “deep miniaturization” manufacturing initiative lasting ten years. Unlike other pacemakers, this revolutionary piece of implantable medical equipment is implanted inside of the heart. Researchers have found that this exceptionally small product is capable of industry-leading performance. When compared to traditional pacing systems, Medtronic’s miniaturized device caused significantly fewer major health complications.
Wearable Health Devices & Miniaturized Components
Wearable health devices allow continuous physiological monitoring, supporting remote care and preventive health care models that greatly depend on patients’ ability to engage in self-monitoring. Miniaturized components and sensors, advanced power management, and high-performance, low-cost display technologies are making the wearable health market more advantageous for OEMs in the medical devices, and consumer electronics sector.
 Standout Innovation | Wearable sweat sensor: This wearable health monitor, designed by engineers at UC Berkeley, is the first fully integrated device capable of providing continuous, non-invasive monitoring of multiple biochemicals in sweat. Their solution’s flexible sensor system selectively measures metabolites and electrolytes in sweat before wirelessly transmitting and syncing this processed data to smartphone devices in real time.
Standout Innovation | Wearable sweat sensor: This wearable health monitor, designed by engineers at UC Berkeley, is the first fully integrated device capable of providing continuous, non-invasive monitoring of multiple biochemicals in sweat. Their solution’s flexible sensor system selectively measures metabolites and electrolytes in sweat before wirelessly transmitting and syncing this processed data to smartphone devices in real time.
Classes Of Medical Devices Being Transformed By Miniaturized Components
- Cochlear implants
- Emergency response safety monitoring systems
- Health and wellness monitoring systems
- Implantable defibrillator
- Implantable cardiac pacemakers
- Implantable neurostimulator systems
- Implantable/programmable infusion pumps
- Implantable continuous glucose monitor (CGM)
- Treatment efficacy assessment
With 01005 packages and other miniaturized components, OEMs are realizing new opportunities by entering into and, in some cases, creating entirely new product markets. Few are engaged in custom chip design to achieve device miniaturization. Most OEMs are using bare dies and flip-mounting them directly onto the substrate, customizing flexible PCBs, or simply purchasing commercially available chips, microcontrollers or analog components.
OEMs should involve their PCB assembly vendors in the product design and development process as early as possible. More traditional OEM-vendor relationships simply aren’t adequate when manufacturing miniaturized devices. PCB assemblers must provide OEMs guidance on design, testing and mass production challenges often confronted when assembling small package sizes. They can also advise OEMs on selecting suppliers with optimal pricing and component availabilities. OEMs that fail to uncover supply chain or logistical and component compatibility challenges early in the concept and design stage risk affecting go-to market costs and timelines in the manufacturing stage. PCBA vendors with specialized equipment and knowledgeable support teams are, in fact, the secret weapon behind OEMs embracing the device miniaturization trend.

